How many times have you evaluated acute, non-traumatic, monoarticular arthritis while contemplating the risks and benefits of arthrocentesis to rule-out septic arthritis? What about ruling-in a crystalloid arthritis like gout?
An evidence-based approach to the evaluation and management of acute single joint pain
How many times have you evaluated acute, non-traumatic, monoarticular arthritis while contemplating the risks and benefits of arthrocentesis to rule-out septic arthritis? What about ruling-in a crystalloid arthritis like gout? Clinicians are able to deduce the etiology of acute nontraumatic joint pain/swelling within 3-days in most cases, but in an era of “overdiagnosis” and “overtreatment” ED providers lack the luxury of a 3-day admission for most monoarticular arthritis patients. ED physicians’ clinical decision making often skews to the “rule out worst case scenario” model, which in the case of an acutely painful/swollen joint includes septic arthritis. In this essay, we intend septic arthritis to include all bacterial synovial fluid infections except gonococcus which we distinguish as a separate disease process with distinct diagnostic testing and treatment approaches. About 50% of septic arthritis cases involve the knee, but any synovial space can be infected. Septic arthritis management options include surgical drainage and systemic antibiotics, although needle aspiration has also been evaluated in select cases. Short-term mortality for treated septic arthritis ranges from 3% to 11% (Cooper 1986, Deesomchok 1990, Kaandorp 1995, Gupta 2001).
In my training, I saw a variety of approaches in the evaluation of acute single joint pain/swelling, although the textbook answer was always that septic arthritis cannot be excluded without synovial fluid analysis – every time on every patient. Of course, I always wondered how accurate history, physical exam, serum labs, and synovial findings were for septic arthritis. Textbooks rarely provided the quantitative estimates of ED-specific prevalence for septic arthritis in this scenario, nor did these tomes of wisdom enlighten me with likelihood ratios for CBC, synovial white blood cell count, gram stain, or synovial cultures. Thanks to the new Evidence Based Diagnostics series in Academic Emergency Medicine, these details are more transparent today. These concepts are increasingly important in an era of specialty-specific “Choosing Wisely” campaigns to contain wasteful spending while concurrently optimizing diagnostic and therapeutic efficiencies in our clinical care protocols.
How Likely Is Septic Arthritis in ED Patients?
The differential diagnosis for acute monoarticular arthritis presenting to the ED is broad and includes infections (bacterial, fungal, mycobacterial, viral), crystalloid arthropathy (gout, pseudogout), rheumatoid arthritis, and trauma. Based upon the sole ED-centric systematic review available on this topic, the best estimate pre-test probability for septic arthritis in the ED is 27%. This does not mean that 27% of ED patients with acute monoarticular pain have septic arthritis. Remember that this estimate is derived from a prospective ED-based study in which the patient first passed through the filter of emergency physician gestalt before being enrolled in the study. In other words, the EPs were specifically concerned about septic arthritis. This means that among all ED patients in whom the clinician believes that septic arthritis is sufficiently likely to merit an arthrocentesis, 27 in 100 will actually have septic arthritis. Most experts agree that this is an overestimate, but there is currently no better estimate of pre-test probability to facilitate Bayesian reasoning.
Can I rely upon history or physical exam to rule-out septic arthritis?
The majority of emergency medicine diagnoses lack significant high-quality diagnostic research to quantify the diagnostic accuracy of history, physical exam, clinical gestalt, or lab testing. Septic arthritis is no exception. In fact, diagnostic studies of history and physical exam to evaluate septic arthritis in any setting are virtually non-existent: two studies assess historical risk factors and none evaluate physical exam. One attainable measure of quality is incorporation of the STARD principles which are publication guidelines for diagnostic studies that ensure internal validity and reproducibility across studies. None of the septic arthritis studies (including the lab test studies) adhere to the STARD criteria in design or intent. Therefore, the available evidence may provide skewed estimates of diagnostic accuracy because of various biases, including spectrum bias, double-gold standard bias, and incorporation bias.
Nonetheless, the available evidence for historical risk factors identified just three factors with a likelihood ratio greater than 3: prosthetic joint with overlying skin infection (positive likelihood ratio [LR+] 15), joint surgery within the preceding 3 months (LR+ 6.9), and age over 80 (LR+ 3.5). The absence of risk factors does not significantly reduce the probability of septic arthritis (the range of negative likelihood ratios [LR-] was 0.64-0.93). As a general rule-of-thumb, a clinically helpful diagnostic test will have a LR+ greater than 10 to rule-in a diagnosis or a LR- less than 0.1 to rule-out a diagnosis.
 Do blood tests help rule-in or rule-out septic arthritis?
Do blood tests help rule-in or rule-out septic arthritis?
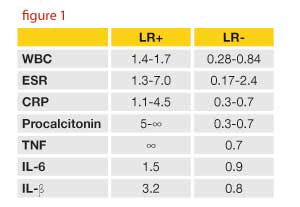
Commonly available serum tests (WBC, ESR, CRP) for septic arthritis are inaccurate and relatively worthless acutely. Eliminating these serum tests from the lexicon of reflexive ED testing for septic arthritis is an attractive target to “Choose Wisely” in reducing unhelpful, potentially expensive laboratory testing. Some orthopedic surgeons believe that serial assessment of ESR and CRP is anecdotally helpful for the longitudinal diagnosis and prognosis for suspected septic arthritis. However, I have never been able to identify any evidence to support this practice. Although serum procalcitonin and tumor necrosis factor demonstrate promise to rule-in the diagnosis (see LR+ ranges in figure 1), these tests are not commonly available in the ED.

How accurate are the synovial white blood cell count and other synovial tests?
The suboptimal diagnostic test characteristics for history, physical exam, and serum tests leaves synovial testing as the standard of care diagnostic strategy to evaluate for septic arthritis. The sole systematic review noted that the risk of arthrocentesis includes post-procedural iatrogenic infections with a range of 0.01% in healthy populations to 0.05% in immunocompromised patients.
How well does the synovial fluid distinguish non-gonococcal bacterial arthritis from other acute joint problems? Synovial gram stain has sensitivity 29% – 65% and no study has assessed specificity (Argen 1966, Goldenberg 1976, McCutchan 1990, Faraj 2002, McGillicuddy 2007). The synovial white blood cell count (sWBC) can be evaluated as a dichotomous test (LR+ and LR- above or below a certain threshold) or as a continuous test (using a concept called interval likelihood ratios). Most textbooks and original research reports use the dichotomous test approach. The AEM Evidence Based Diagnostic meta-analysis summary estimates for dichotomous LRs at various thresholds are noted in figure 2. On the other hand, a synovial WBC > 100,000 cells/mm3 has an interval LR of infinity, whereas synovial WBC 0-25, 25-50, and 50-100 yield interval LR of 0.33, 1.06, and 3.59, respectively.
So how do we tackle all these numbers in order to make a quantitative estimate of septic arthritis at the bedside? This is where the concept of Bayesian reasoning comes in. Just start with an estimate of septic arthritis probability before ordering any lab tests (your pre-test or prior probability). We’ll use 27%, derived from this evidence based diagnostic synopsis. Next, use online calculators to compute post-test probabilities once your lab results are available.
You might notice that you’ve got a couple of ways of doing the calculation. If your tap reveals a synovial WBC count of 30,000 cells / mm3, you could use the LR+ for >25,000 or the LR- for 25 threshold, the post-test probability becomes 54%. If using the <50 threshold, the post-test probability becomes 16%.
On the other hand, using interval LRs there is only one correct diagnostic accuracy estimate (iLR25-50 = 1.06) and no philosophical arguments about which value to use. The iLR approach would yield a post-test probability of 28% if the synovial WBC was between 25k and 50k. The interval LRs from the Evidence Based Diagnostic synopsis – and their impact on pre-test probability – are shown in figure 3.
Are there any new tests for septic arthritis?
Synovial lactate is a promising test for the ED evaluation of suspected acute septic arthritis. Unfortunately, two of the three studies did not distinguish whether they assessed D- or L-lactate! Bacteria, not humans (with rare exceptions such as short gut syndrome), produce D-lactate. On the other hand, humans, not bacteria, produce L-lactate. Laboratory Medicine experts believe that D-lactate is biologically plausible as a diagnostic marker of bacterial arthritis, but no commercially available laboratory test currently exists for D-lactate. Instead, D-lactate is a 3-day mail out test. On the other hand, it is conceivable that L-lactate is correlated with the synovial white blood cell count with higher sWBC counts producing more L-lactate. This hypothesis has not been tested. In fact, none of the synovial lactate studies evaluated the L- or D-lactate in real-time. Instead, specimens were frozen and tested in batches long after the patient care episode was past. Nonetheless, when contemplating the diagnostic accuracy of sWBC and gram stain, synovial lactate is promising. Here is the study-by-study synopsis of the available data.
Gratacós 1995
This is the only study to specify which form of lactate they assessed (D-lactate). Interval LR’s range from 0.16 (D-lactate 0-0.05 mmol/L) to 20 (D-lactate >0.15 mmol/L). This test is superior to synovial WBC > 50,000 cells/mm3 (LR+ 9.3, LR- 0.47) or sPMN > 90% (LR+ 2.7, LR- 0.37) and may be particularly useful for partially treated septic arthritis.
Brook 1978
At a threshold of 5.5 mmol/L (note 10-fold higher than Gratacós’ study which may indicate that Brook was studying L-lactate) the synovial lactate LR+ is 5.9 and LR- 0.04. More importantly, the interval LR for 0-5.5 mmol/L is 0.06 and for >16.7 mmol/L it is infinity. Unfortunately, the authors do not describe whether they evaluated D-lactate or L-lactate and by failing to adhere to STARD criteria, the authors leave open the possibility for significant biases, most of which skew estimates of sensitivity and specificity upwards.
Moss 1981
Based on this case-control, single-center, Rheumatology clinic study, synovial lactate assays (probably L-lactate based on the investigator’s discussion) using the Calbiochem-Behring Rapid Lactate Kit accurately discriminates non-GC septic arthritis from other etiologies of acute monoarticular joint pain/swelling. At a threshold of 10 mmoL/L (which is nearly two-fold the 5.5 mmoL/L threshold proposed by Brook 1978) the LR+ is 22 and the LR- is 0. Using the Brook 5 mmoL/L threshold, Moss demonstrates LR+ 2.7 and LR- 0 (compared with 5.9 and 0.04 for Brook, respectively) for lactate. The interval LR for 0-10 mmol/L is zero versus 17.2 for 10-20 mmoL/L and infinity for synovial lactate >20 mmoL/L.
What’s the Bottom Line?
In several studies, synovial lactate levels > 10 mmol/L levels consistently ruled-in septic arthritis. While no studies have yet used point-of-care lactate assays or studied the effect on ED management of septic arthritis, the impact of point-of-care serum lactate on ED sepsis management suggests a compelling need for further research. Future studies should assess diagnostic accuracy in a consecutive sampling of ED patients with monoarticular arthritis in whom there is sufficient suspicion of non-GC septic arthritis to perform an arthrocentesis. It will be essential for these future studies to follow the STARD criteria and to report interval LR’s. If these “level 2” diagnostic accuracy studies confirm synovial lactate as a useful adjunct to synovial WBC, the logical progression in research would be to assess the impact of awareness of synovial lactate on clinician decision-making. For now, maintain an appropriate threshold for arthrocentesis in suspected septic arthritis and interpret synovial results with appropriate diagnostic prowess understanding exactly how accurate or inaccurate these tests are in moving you towards or away from this important disease. Keep an eye out for more septic arthritis original research!
Chris is an associate professor of emergency medicine and the Director of Evidence Based Medicine at Washington University in St. Louis. He is also the current Chair of the SAEM Evidence Based Medicine Interest Group and also Chair of the ACEP Geriatric Section. He is co-author of the textbook “Evidence-Based Emergency Care: Diagnostic Testing and Clinical Decision Rules, 2nd Edition”.